1.5 THE BASIC ELEMENTS OF A LIQUID PROPELLANT ROCKET ENGINE SYSTEM
A vehicle system has occasionally been defined as a purposeful conglomeration of subsystems. One of these is the engine system. The definition of the scope of the various vehicle subsystems has not always been uniform and probably, by necessity, never will be. For instance, for vehicle systems in which the propellant tanks simultaneously serve as the vehicle airframe, it may be a matter of opinion whether they are part of the structure or of the engine system. The decision to which subsystem they belong may well depend on the fact whether the tanks will be supplied by the engine manufacturer, or by a separate contractor. Similarly, some, notably the engine system supplier, may consider the guidance system a part of the payload, while the vehicle user will hold that anything without which the vehicle cannot fly reliably and accurately to its destination is not payload. Whatever the definitions may be, it is important that they are used uniformly and consistently in a given project.
For the purpose of this book, we will define a vehicle as being composed of the following major subsystems: (1) Engine system (2) Vehicle structure (3) Guidance system (4) Payload (5) Accessories
In the following, we will concern ourselves with the engine system only, except for brief references to the other systems, as required. We
Table 1-4.-General Data of Some Storable Liquid Rocket Propellants
| Propellant | Formula | Use | Mol. wt. | Freezing point | Boiling point | Vapor press., psia | Density gm/cc | Stability | Handling hazard | Storability | Materials compatibility | Cost $/lb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aniline | Fuel. coolant | 93.2 | 21 | 364 | 0.25 at | 1.022 at | Good | Good | Good | Al., steel, Teflon, Kel-F | ........ | |
| Bromine pentafluoride | Oxid., coolant | 174.9 | -80.5 | 104.5 | 41 at | 2.48 at | Up to | Reacts with fuel | Good | Al. alloy. 18-8 stainless steel, nickel alloy. copper, Teflon | 4.75 | |
| Chlorine trifluoride | Oxid. | 92.5 | -105.4 | 53.15 | 80 at | 1.825 at | Up to | Toxic | Good below | Al. alloy. 18-8 stainless steel. nickel alloy. copper, Teflon | 0.50-2.50 | |
| 92.5% E.A. (ethyl alcohol) | Fuel, coolant | 41.25 | -189 | 172 | 13 at | 0.81 at | Good | Flammable | Good below | Al., steel, nickel alloy, Teflon. Kel-F, polyethylene | 0.15 | |
| Hydrazine | Fuel. oxid., coolant | 32.05 | 34.5 | 235.4 | 2.8 at | 1.01 at | Up to | Toxic, flammable | Good | Al., 304.307 stainless steel, Teflon, Kel-F, polyethylene | 0.50-3.00 | |
| 95% hydrogen peroxide | Monoprop., oxid.. coolant | 32.57 | 21.9 | 294.8 | 0.05 at | 1.414 at | Unstable decomp. at | Hazardous skin contact. flammable | Deteriorates at . | Al., stainless steel. Teflon, Kel-F | 0.50 | |
| 98% hydrogen peroxide | Same as above | 33.42 | 27.5 | 299.2 | 0.043 at | 1.432 at | Same as above | Same as above | Same as above | Same as above | 1.00 | |
| Hydyne (40% "Deta" 60% "UDMH") | , | Fuel, coolant | 72.15 | 65 | 140 to 400 | 16.5 at | 0.855 at | Good | Toxic | Good | Al., stainless steel, Teflon, Kel-F | 0.50-2.00 |
| IRFNA (inhibited red fuming nitric acid) | 82% . . 2% , 1% HF | Oxid.. coolant | 55.9 | -57 | 150 | 17.3 at | 1.57 at | Good | Toxic, hazardous skin contact | Good | Al., stainless steel. Teflon, Kel-F, polyethylene | 0.08-0.10 |
| JP-4 (jet propulsion fuel) | Fuel, coolant | 128 | -76 | 270 to 470 | 7.2 at | 0.747 to 0.825 | Good | Vapor explosive | Good | Al.. steel, nickel alloy, neoprene, Teflon, Kel-F | 0.015 | |
| MMH (monomethylhydrazine) | Fuel, coolant | 46.08 | -63 | 187 | 8.8 at | 0.878 at | Good | Toxic | Good | Al., 304.307 stainless steel, Teflon. Kel-F, polyethylene | 0.62-6.25 |
Table 1-4.-General Data of Some Storable Liquid Rocket Propellants (Continued)
| Propellant | Formula | Use | Mol. wt. | Freezing point | Boiling point | Vapor press.. psia | Density | Stability | Handling hazard | Storability | Materials compatibility | Cost $/lb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitrogen tetroxide | Oxid. | 92.02 | 11 | 70 | 111 at | 1.44 at | Function of temp. | Very toxic, hazardous skin contact | Good when dry | Al., stainless steel, nickel alloy. Teflon | 0.075 | |
| Pentaborane | Fuel | 63.17 | -52.28 | 140.11 | 19 at | 0.61 at | Good | Explosive on exposure to air, very toxic | Good | Al., steel, copper, Teflon, Kel-F. Viton A | 2.50-5.00 | |
| Propyl nitrate | Fuel, coolant | 105.09 | -130.9 | 231 | 3.7 at | 1.06 at | Fair | Sensitive to shock | Good | Al., stainless steel, Teflon, Kel-F | ....... | |
| RP-1 (rocket propellant) | Mil-Spec.F25576B | Fuel, coolant | 165 to 195 | -47 to -64 | 342 to 507 | 0.33 at | 0.8 to 0.82 at | Auto. ignition at | Flammable | Good | Al., steel, nickel alloy, copper. Teflon, Kel-F. Neoprene | 0.015 |
| TEA (triethylaluminum | Fuel, start compound | 114.15 | -49.9 | 381 | 0,40 at | 0.836 at | Decomp. over | Ignites on contact with air | Good | Al., steel, copper, Teflon | ....... | |
| TMA (trimethylamine) | Fuel | 59.11 | -179 | 37 | 108 at | 0.603 at | Good | Good | Good | Al., steel, copper, Teflon | ........ | |
| TMB-1, 3-D ( 'N'-tetra-methylbutane-1. 3-diamine) | Fuel. coolant | 144.2 | -131 | 320 | 1.32 at | 0.795 at | Stable 1 hr. at | Good | Al., 347 stainless steel, polyethylene | ....... | ||
| TNM (tetranitromethane) | Oxid. | 196.04 | 57.3 | 259 | 2.38 at | 1.64 at | Thermal. unstable | Shock sensitive | Good below | Al., mild steel, Teflon, Kel-F | 0.30 | |
| UDMH (unsymmetrical dimethylhydrazine) | Fuel. coolant | 60.08 | -72 | 146 | 17.6 at | 0.789 at | Good | Toxic | Good | Al., stainless steel, Tefion, Kel-F | 0.50-2.00 | |
| WFNA (white fuming nitric acid) | 97.5% HNO. , | Oxid., coolant | 59.9 | -45 | 186 | 9.09 at | 1.46 to 1.52 at | Decomp. above | Toxic, hazardous skin contact | Fair | Al., stainless steel, Teflon, Kel-F, polyethylene | 0.15 |
Table 1-5.-General Data of Some Cryogenic Liquid Rocket Propellants
| Propellants | Formula | Use | Mol. wt. | Freezing point. | Boiling point. | Critical press., psia | Critical temp., | Density at boiling point | Stability | Handle hazard | Materials compatibility | Cost $/lb | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| - A B Bietensencis | Anmmonia ......... | Fuel, coolant | 17.03 | 108 | -28 | Vapor pressure = 500 psia at | 0.683 | Good | Toxic, flammable | Al., steel, lead, Tetlon, Kel-F. Vitron A | 0.04 | ||
| Liquid fluorine | Oxid. | 38.00 | -364 | -307 | 808 | -200.5 | 1.509 | Good | Very toxic. flammable | Al., 300 series stainless steel, nickel alloy, brass | 6.00 | ||
| Liquid hydrogen | Fuel, coolant | 2.016 | -434.6 | -422.9 | 187.8 | -400.3 | 0.071 | Good | Flammable | Stainless steel, nickel alloy. Al alloy, Kel-F | 7.00 | ||
| Liquid oxygen. | Oxid. | 32.00 | -362 | -297.4 | 735 | -182 | 1.142 | Good | Good | Al. . stainless steel. nickel alloy, copper. Teflon, Kel-F | 0.05 | ||
| Oxygen difluoride. . | Oxid. | 54.00 | ....... | -299 | 719 | -72.3 | 1.521 | Good | Very toxic, flammable | Al., 300 series stainless steel. nickel alloy, brass | .... | ||
| Ozone | Oxid. | 48.00 | -420 | 168 | 804 | 10.2 | 1.46 | Above 20% explosive | Very toxic, flammable | Al. 300 series stainless steel, Teflon, Kel-F |
SENIONG 1ヨYSOd INV77ヨdOdd AINOII NOISEQ
Table 1-6.-Performance of Some Liquid Rocket Monopropellants
| Propellant | Specific impulse , | Density impulse , sec gm/cc | Applications | Remarks |
|---|---|---|---|---|
| Hydrogen peroxide ( ) (95%) . | 140 | 198 | Gas generators for turbopump and auxiliary drive; small control rockets | Difficult handling |
| Hydrazine ( )............. | 205 | 207 | Gas generators: small control rockets | Difficult handling (can decompose at high temperature) |
| Nitromethane ( ) | 180 | 204.8 | Small ordnance rockets | Dangerous handling (can detonate unexpectedly) |
| Methylacetylene. . . . . . . . . . . . . | 160 | 108.6 | Gas generators; small rockets | Safe handling; dangerous and very smoky exhaust fumes |
Theoretical value at . sea-level optimum expansion, frozen gas composition or frozen equilibrium.
TABLE 1-7.-Theoretical Performance of Some Medium-Energy Storable Liquid Rocket Bipropellant Combinations
| Oxidizer | Fuel | 形 | Applications | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| IRFNA ( ) | UDMH | 2.99 | 1.51 | 1.26 | 5340 | 23.7 | 5490 | 1.619 | 276 | 348 | Small air-to-air, |
| 3.24 | 1.63 | 1.27 | 5315 | 24.2 | 5435 | 1.630 | 275 | 350 | arr-to-surface | ||
| Hydrazine | 1.47 | 95 | 1.28 | 5090 | 20.8 | 5690 | 1.602 | 283 | 362 | rockets and | |
| 1.54 | 99 | 1.29 | 5100 | 21.1 | 5665 | 1.608 | 283 | 365 | upper stages of space vehicles | ||
| 50% UDMH-50% hydrazine. | 2.20 | 1.26 | 1.27 | 5250 | 22.4 | 5580 | 1.610 | 279 | 354 | ||
| 2.42 | 1.39 | 1.29 | 5220 | 23.0 | 5510 | 1.618 | 277 | 358 | |||
| Hydyne | 3.11 | 1.70 | 1.31 | 5295 | 24.1 | 5425 | 1.620 | 273 | 358 | ||
| 3.33 | 1.82 | 1.32 | 5270 | 24.5 | 5375 | 1.630 | 272 | 359 | |||
| RP-1. | 4.80 | 2.48 | 1.35 | 5355 | 25.8 | 5275 | 1.636 | 268 | 362 | ||
| 5.14 | 2.65 | 1.36 | 5330 | 26.2 | 5225 | 1.646 | 267 | 363 | |||
| TMB-1, 3-D | 4.09 | 2.08 | 1.32 | 5325 | 25.1 | 5335 | 1.632 | 270 | 356 | ||
| 4.37 | 2.23 | 1.33 | 5300 | 25.5 | 5280 | 1.640 | 269 | 358 | |||
| JP-X (60% JP-4, 40% UDMH) | 4.13 | 2.16 | 1.33 | 5310 | 24.6 | 5320 | 1.628 | 269 | 358 | ||
| 92.5% E.A. | 2.89 | 1.47 | 1.26 | 4935 | 5130 | 1.626 | 259 | 326 | |||
| MMH | 2.47 | 1.38 | 1.28 | 5290 | 5550 | 1.618 | 279 | 357 | |||
| TMA | 4.01 | 1.61 | 1.21 | 5285 | 5375 | 1.625 | 271 | 328 | |||
| 95% hydrogen | UDMH | 4.54 | 2.53 | 1.24 | 4800 | 21.7 | 5530 | 1.620 | 278 | 345 | Manned aircraft, |
| peroxide | 4.74 | 2.64 | 1.25 | 4780 | 21.3 | 5505 | 1.620 | 277 | 346 | small air-to-air. | |
| Hydrazine | 2.17 | 1.54 | 1.26 | 4675 | 19.5 | 5655 | 1.604 | 282 | 355 | air-to-surface | |
| 2.20 | 1.57 | 1.26 | 4675 | 19.5 | 5655 | 1.604 | 282 | 355 | rockets, and | ||
| 50% UDMH-50% Hydrazine | 3.35 | 2.12 | 1.25 | 4760 | 20.5 | 5580 | 1.610 | 279 | 349 | upper stages of | |
| 3.47 | 2.20 | 1.26 | 4740 | 20.6 | 5560 | 1.615 | 279 | 351 | space vehicles | ||
| Hydyne | 4.68 | 2.83 | 1.27 | 4765 | 21.3 | 5485 | 1.622 | 276 | 350 | ||
| 4.87 | 2.95 | 1.28 | 4745 | 21.4 | 5465 | 1.619 | 275 | 352 | |||
| RP-1 | 7.35 | 4.18 | 1.30 | 4785 | 22.1 | 5405 | 1.627 | 273 | 355 | ||
| 7.58 | 4.32 | 1.31 | 4765 | 22.2 | 5390 | 1.620 | 271 | 355 | |||
| TMB-1, 3-D | 6.20 | 3.49 | 1.28 | 4770 | 218 | 5440 | 1622 | 274 | 351 | ||
| 6.45 | 3.63 | 1.29 | 4745 | 21.9 | 5415 | 1.618 | 272 | 351 |
Table 1-7.-Theoretical Performance of Some Medium-Energy Storable Liquid Rocket Bipropellant Combinations (Continued)
| Oxidizer | Fuel | d | 刃 | c* | Applications | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitrogen tetroxide | UDMH | 2.95 | 1.61 | 1.20 | 5685 | 24.5 | 5555 | 1.632 | 282 | 339 | Manned aircraft, |
| Hydyne | 2.71 | 1.61 | 1.22 | 5650 | 24.1 | 5580 | 1626 | 282 | 344 | ICBM, IRBM, | |
| 2.95 | 1.75 | 1.24 | 5655 | 24.7 | 5525 | 1.631 | 280 | 347 | ALBM, small air- | ||
| RP-1 | 4.04 | 2.26 | 1.25 | 5745 | 25.7 | 5440 | 1.636 | 276 | 345 | to-air rockets. upper stages of space vehicles | |
| 4.50 | 2.51 | 1.27 | 5755 | 26.5 | 5385 | 1.639 | 274 | 348 | |||
| TMB-1, 3-D | 3.55 | 1.96 | 1.23 | 5715 | 25.2 | 5495 | 1631 | 278 | 342 | ||
| 3.90 | 2.15 | 1.24 | 5710 | 25.9 | 5425 | 1.645 | 277 | 344 | |||
| 925%E.A. | 2.59 | 1.45 | 1.19 | 5290 | 5260 | 1.635 | 267 | 318 | |||
| Chlorine trifluoride | UDMH | 3.03 | 1.31 | 1.38 | 6305 | 25.8 | 5630 | 1.602 | 280 | 386 | ICBM, IRBM. |
| 3.28 | 1.42 | 1.40 | 6330 | 26.2 | 5605 | 1.589 | 277 | 388 | ALBM, and small air-launched rockets, upper stages of space vehicles | ||
| Hydvne RP-1 | 298 | 140 | 1.43 | 6220 | 26.1 | 5555 | 1.599 | 276 | 395 | ||
| 320 | 1.50 | 1.44 | 6250 | 26.5 | 5535 | 1.595 | 274 | 395 | |||
| 3.20 | 142 | 141 | 5890 | 29.1 | 5140 | 1.618 | 258 | 364 | |||
| 12.80 | 5.66 | 1.68 | 5735 | 370 | 4535 | 1.636 | 230 | 386 | |||
| TMB-1, 3-D | 3.17 | 1.39 | 1.40 | 6035 | 27.6 | 5330 | 1.608 | 266 | 373 | ||
| 3.60 | 1.57 | 143 | 6040 | 28.1 | 5280 | 1.592 | 261 | 374 | |||
| Bromine pentafluoride | Hydrazine | 3.35 | 1.37 | 1.86 | 5570 | 5000 | 1.565 | 243 | 453 | Small air-launched rockets |
Table 1-8.-Theoretical Performance of Some High-Energy Storable Liquid Rocket Bipropellant Combinations
| Oxidizer | Fuel | d | c* | Applications | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydrogen peroxide | Hydrazine | 2.01 | 1.41 | 1.26 | 4775 | 19.5 | 5735 | 1.601 | 285 | 359 | ICBM, IRBM, |
| Pentaborane | 2.70 | 1.188 | 1.037 | 5390 | 19.01 | 6067 | 1.600 | 302 | 313 | ALBM | |
| Nitrogen tetroxide | UDMH | 2.61 | 1.42 | 1.18 | 5685 | 23.6 | 5650 | 1.624 | 285 | 336 | FBM, ICBM. |
| Hydrazine | 1.34 | 93 | 1.22 | 5390 | 20.9 | 5845 | 1.610 | 292 | 357 | IRBM. ALBM. | |
| 1.42 | 99 | 1.23 | 5415 | 21.3 | 5815 | 1.605 | 290 | 357 | upper stages | ||
| 50% UDMH-50% Hydrazine | 2.00 | 1.24 | 1.21 | 5590 | 22.6 | 5725 | 1.620 | 288 | 348 | of space | |
| 2.15 | 1.33 | 1.21 | 5570 | 23.0 | 5665 | 1.636 | 288 | 348 | vehicles | ||
| MMH. . | 2.16 | 131 | 1.20 | 5635 | 5720 | 1.621 | 288 | 346 | |||
| Chlorine trifluoride | Hydrazine | 2.77 | 1.53 | 1.51 | 6550 | 232 | 5995 | 1582 | 294 | 444 | FBM, ICBM, |
| 2.94 | 1.62 | 1.52 | 6600 | 23.6 | 5980 | 1.572 | 292 | 444 | IRBM, ALBM. | ||
| 50% UDMH-50% Hydrazine | 2.89 | 1.42 | 1.45 | 6385 | 24.5 | 5795 | 1596 | 287 | 416 | upper stages | |
| 3.11 | 1.53 | 1.46 | 6420 | 24.9 | 5770 | 1.598 | 286 | 417 | of space | ||
| MMH | 3.00 | 1.44 | 1.44 | 6400 | 5763 | 1.591 | 285 | 410 | vehicles | ||
| Hydrazine | Pentaborane | 1.4 | 85 | 796 | 4430 | 14.7 | 6402 | 1.644 | 327 | 261 | ICBM. IRBM |
TABLE 1-9.-Theoretical Performance of Some High-Energy Cryogenic Liquid Rocket Bipropellant Combinations
| Oxidizer | Fuel | Iv | d | c* | Applications | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liquid oxygen | RP-1................... | 2.00 | 1.421 | 0.998 | 5760 | 211 | 5898 | 1.605 | 294 | 293 | ICBM, IRBM, large space-probe and space craft boosters |
| 2.40 | 1.708 | 1.012 | 6100 | 22.8 | 5953 | 1.620 | 300 | 303 | |||
| 2.56 | 1.82 | 1.02 | 6150 | 23.3 | 5920 | 1.632 | 300 | 306 | |||
| 2.73 | 1.94 | 1.03 | 6200 | 239 | 5865 | 1.642 | 299 | 308 | |||
| Ammonia | 1.30 | . 78 | 88 | 5055 | 19.3 | 5920 | 1.608 | 296 | 260 | ||
| 1.40 | 84 | 89 | 5100 | 198 | 5865 | 1.612 | 294 | 261 | |||
| 95% E.A. | 1.73 | 1.23 | 99 | 5640 | 24.1 | 5605 | 1.648 | 287 | 284 | ||
| 1.80 | 1.28 | 1.00 | 5675 | 24.4 | 5585 | 1.644 | 285 | 285 | |||
| Hydrazine | . 90 | 80 | 1.07 | 5660 | 19.3 | 6235 | 1.618 | 313 | 335 | ||
| 50% UDMH-50% Hydrazine | 1.30 | 1.03 | 1.02 | 5980 | 20.6 | 6160 | 1.628 | 312 | 318 | ||
| 1.37 | 1.08 | 1.03 | 5905 | 20.9 | 6155 | 1.629 | 310 | 319 | |||
| Hydyne | 1.73 | 1.31 | 1.02 | 5990 | 21.8 | 6035 | 1.632 | 306 | 312 | ||
| 1.80 | 1.36 | 1.02 | 6030 | 22.2 | 6010 | 1.639 | 306 | 312 | |||
| UDMH | 1.65 | 1.14 | 98 | 6010 | 21.3 | 6115 | 1.631 | 310 | 30.4 | ||
| 1.83 | 1.27 | 99 | 6065 | 22.1 | 6040 | 1.638 | 307 | 304 | |||
| TMB-1.3-D | 2.28 | 1.60 | 1.01 | 6100 | 22.9 | 5945 | 1.642 | 303 | 306 | ||
| 2.37 | 1.66 | 1.01 | 6120 | 23.2 | 5915 | 1.650 | 303 | 306 |
Table 1-10.-Theoretical Performance of Some Very-High-Energy Cryogenic Liquid Rocket Bipropellant Combinations
| Oxidizer | Fuel | d | m | c* | Applications | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liquid oxygen | Liquid hydrogen | 4.02 | 0.25 | 0.28 | 4935 | 10.0 | 7980 | 1.578 | 391 | 109 | Space probe and space craft upper stage and booster |
| 19.50 | 1.20 | 65 | 4960 | 23.4 | 5300 | 1.610 | 265 | 172 | |||
| Liquid fluorine | Hydrazine | 2.30 | 1.54 | 1.31 | 7955 | 19.4 | 7245 | 1.615 | 363 | 476 | Space probe upper stage |
| 2.40 | 1.61 | 1.32 | 7980 | 19.6 | 7225 | 1.614 | 362 | 478 | |||
| Liquid hydrogen. . 7.60 | 35 | 45 | 6505 | 11.8 | 8365 | 1.578 | 410 | 185 | |||
| Ammonia | 23.70 | 1.10 | 82 | 8230 | 18.5 | 7515 | 1.592 | 372 | 305 | ||
| 3.29 | 1.48 | 1.18 | 7715 | 19.3 | 7155 | 1.605 | 357 | 421 | |||
| 3.40 | 1.53 | 1.18 | 7745 | 19.5 | 7140 | 1.612 | 357 | 422 |
NOTES FOR TABLES 1-7 THROUGH 1-10
(1) Conditions upon which the performance calculations are based = (a) Combustion chamber pressure (b) Nozzle exit pressure = ambient pressure = 14.7 psia (optimum nozzle expansion ratio at sea-level operation) (c) Chamber contraction ratio (chamber area/nozzle throat area) = infinity (d) Adiabatic combustion (e) Isentropic expansion of ideal gas with shifting composition or shifting equilibrium in the nozzle (2) Symbols: Propellant weight mixture ratio (wt. oxidizer/wt. fuel) Propellant volume mixture ratio (vol. oxidizer/vol. fuel) d = Bulk density of propellant combination (gm/cc). (The density at boiling point was used for those oxidizers or fuels which boil below at one atmosphere pressure) Theoretical chamber temperature, Average molecular weight of combustion products at Theoretical characteristic velocity (ft/sec)
NOTES FOR TABLES 1-7 THROUGH 1-10 (Continued)
Theoretical thrust coefficient Theoretical maximum specific impulse, lb-sec/lb Theoretical maximum density impulse. sec-gm cc (3) To approximate and at other chamber pressures.
Pressure (psia): | Multiply by- |
| ---: | | |
further define that the engine system shall comprise all parts without which the propulsive force cannot be generated. Thus, we will include the propellant tanks and their accessories. A system thus defined frequently is called a propulsion system. We know, from the above, that by including the tanks, we may be "infringing" on the vehicle structure by other definitions.
Thus prepared, we may now proceed to subdivide the engine system further into major components or subassemblies as follows: (1) Thrust chamber assembly (2) Propellant feed system: One of the following two is generally used: Pressurized gas propellant feed system and turbopump propellant feed system. The latter includes some type of tank pressurization system (3) Valves and control systems (4) Propellant tankage (5) Interconnect components and mounts
Depending on the engine system selected, one or another subsystem may not be required or may be integrated with another one. Typical liquid propellant rocket engine systems are shown in figures 1-12 and 1-13.
The rocket has occasionally been called the simplest propulsion system known. The simplest form of a solid propellant rocket or of a pressurized gas-fed storable liquid propellant rocket appears to come close to this ideal. Unfortunately, simplicity frequently is synonymous with inflexibility. Due to vehicle requirements, substantial departures from the basic simplicity may become necessary to meet requirements such as: light weight, high performance, thrust control, thrust vector control, restartability, cutoff im-
| 900 | 99 |
|---|---|
| 800 | 98 |
| 700 | 97 |
| 600 | . 95 |
| 500 | 93 |
| 400 | 91 |
| 300 | 88 |
pulse control, propellant utilization control (sometimes called propellant management), storability, ease of handling, etc. Thus, modern rocket engines contain more subsystems than their basic principle of operation may suggest, to meet the often stringent vehicle requirements. This is true for both liquid as well as solid propellant systems. In general however, the liquid propellant engine is the more flexible one, particularly where large systems are considered.
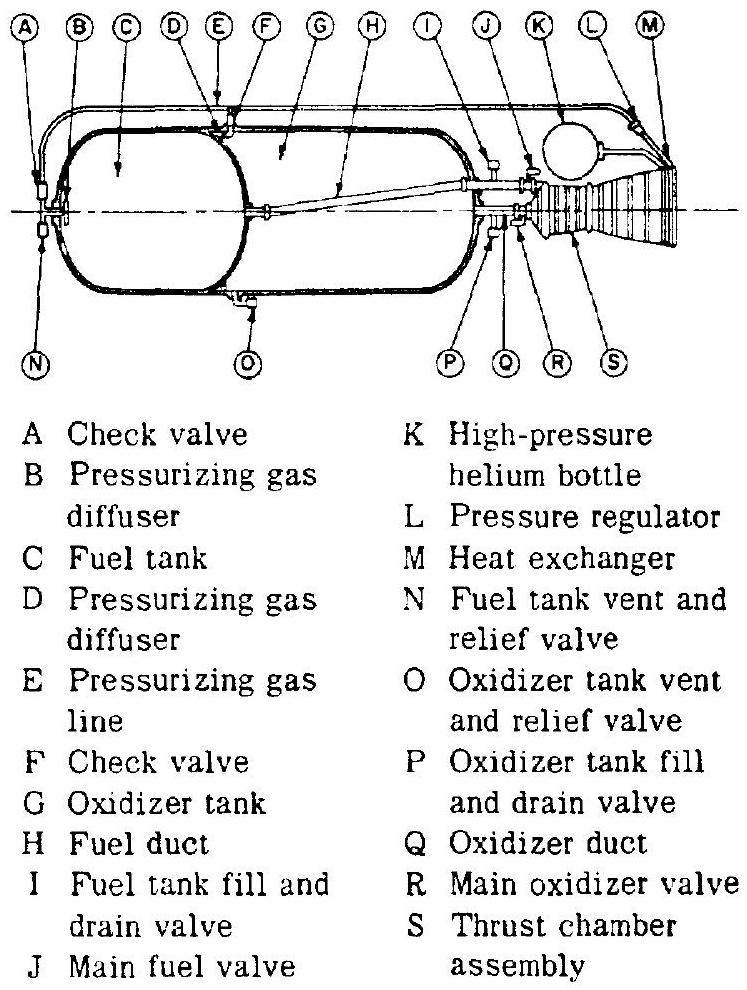 Figure 1-12.-Typical pressurized gas feed liquid propellant rocket engine system.
Figure 1-12.-Typical pressurized gas feed liquid propellant rocket engine system.
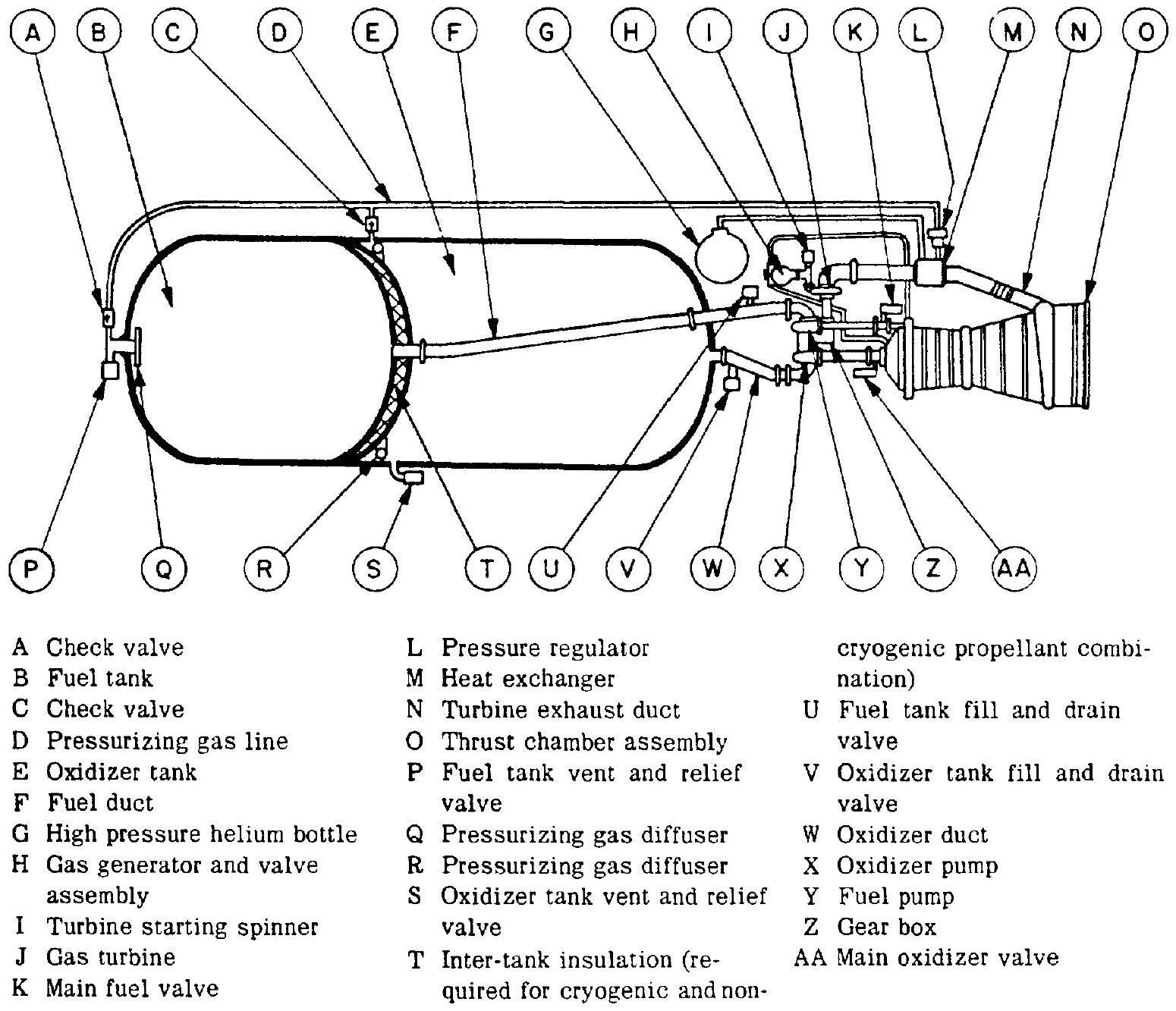 Figure 1-13. - Typical turbopump feed liquid propellant rocket engine system.
Figure 1-13. - Typical turbopump feed liquid propellant rocket engine system.